5.2.2 Results
The RSD was 0.78 % within a day and 1.56% within three days,
which complies with the USP 24 requirements (RSD should be less than 2%).
The results of the drug content (Table 5.1) show that the
S&R formulation did not comply with the USP 24 specifications for
paracetamol content (90% - 110% of the labelled amount) after 6 months of
storage under simulated tropical conditions (40°C and 75 % RH).
Table 5.1: The paracetamol content (expressed
as percentage of the labelled amount) before and after 6 months of storage at
40°C and 75 % RH.
Manufacturer
% of the labelled amount per tablet
0 months 6 months
Regal Pharmaceuticals (Cetamol) 95.6
92.5
Smith Kline Beecham (Panadol) 95.0
93.8
S&R Pharmaceuticals (Saramol) 90.8
87.7
II.5.3 In vitro dissolution
5.3.1 Methods
· Preparation of dissolution
medium
68 g of potassium dihydrogen phosphate was diluted to 10.0 L
with distilled water. The pH was adjusted to 5.8 0.05 using a 0.2 N solution
of sodium hydroxide.
· Calibration curve
Stock solution
200 mg of accurately weighed paracetamol was dissolved in
mobile phase to obtain 100.0 ml. 10 ml from this solution were diluted to 100.0
ml. The resulting solution had a concentration of 200 mg/l. 20 ml of this
solution was diluted to 100.0 ml to give a stock solution with a concentration
of 40 mg/l.
Standard solutions
1, 2, 5, 8 and 10 ml of the stock solution were separately
diluted to 10.0 ml using the dissolution medium to obtain standard solutions
having concentrations of 4, 8, 20, 32 and 40 mg/l, respectively.
A calibration curve (absorbance vs. concentration) y = 0.0633x
+ 0.0294 with a correlation coefficient (R2) of 0.9999 was
constructed.
· Dissolution testing
Dissolution profiles were determined using the USP paddle
method (Method 2). Each of 6 tablets was placed inside a dissolution vessel
filled with 900 ml of dissolution medium maintained at 37 0.5°C and
rotated at a speed of 50 rpm.
At different time intervals (5, 10, 15, 20, 25 and 30 min) 5 ml
filtered samples were manually withdrawn, diluted with dissolution medium
(1:40) and spectrophotometrically analyzed at 243 nm. From the absorbance of
each sample, the drug concentration was calculated by means of the calibration
curve.
5.3.2 Results
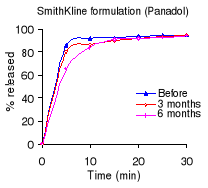
Table 5.2 shows the percentage dissolved within 30 minutes of
dissolution testing and Figure 5.1 the different dissolution profiles. Before
stability testing all drugs complied with the USP 24 dissolution requirements
(not less than 80% of the labelled amount should dissolve within 30 minutes).
The amount of drug released after 10 minutes of dissolution test was more than
80% for the Cetamol and Panadol formulations. The Saramol formulation
disintegrated into larger particles compared to the others two formulations.
The accelerated stability testing did not affect the Cetamol and Panadol drug
percentage released. The amount of drug released from Saramol formulation
decreased, however it remained within USP 24 tolerance limits for dissolution
testing.
Table 5.2: Percentage of paracetamol dissolved within 30
minutes of dissolution testing before and after 3 and 6 months of storage at
40°C and 75% RH. USP requirements: more than 80 % released within 60
minutes.
Manufacturer
% of the labelled amount per tablet
0
months 3 months 6 months
Regal Pharmaceuticals (Cetamol) 95.6
94.6 90.4
Smith Kline Beecham (Panadol) 95.0
94.9 93.9
S&R Pharmaceuticals (Saramol) 90.8
90.1 84.3

Figure 5.1: Dissolution profiles of paracetamol formulations
before and after 3 and 6 months of storage at 40°C and 75 % RH.

| 


