II.1) GENERAL OVERVIEW
The World Health Organization (WHO) recognizes Neisseria
meningitidis, in most countries to be the leading cause of meningitis and
fulminant septicemia and is also recognized to be a significant public health
problem [12] However, large recurring epidemics affect an
extensive region of sub-Saharan Africa known as the « THE MENINGITIS BELT
» (Figure 1) which comprises of 26 countries from Senegal in the West to
Ethiopia in the East [13].

Figure 1: Meningitis Belt in West Africa [14]
Most meningitis cases and out breaks in the African meningitis
belt occur during the epidemic season which tend to extend from November to
June depending on the region [13], with sub-Saharan region
having the world's greatest disease burdens of Haemophilus influenzae
type b streptococcus pneumoniae, and Neisseria meningitides
[15]. An enhanced meningitis surveillance regional
network is also available where the 23 countries participating (Figure 2)
reported in 2017 a total of 29827 of suspected cases of meningitis including
2276 deaths [13]. This was said to represent an increased
5
number of cases compared with 2016 of 18178 suspected cases
resulting also in an increased number of epidemic districts from 42 in 2016 to
57 in 2017[13]
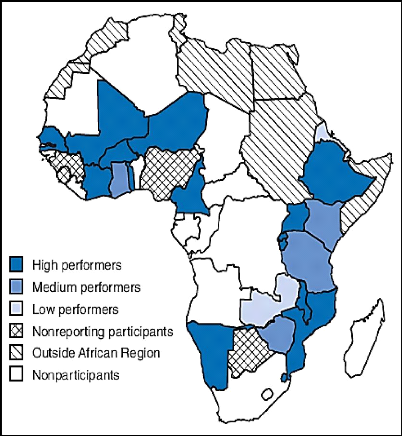
Figure 2: Countries trained to conduct surveillance
for the Pediatric Bacterial Meningitis Surveillance Network, by performance
level* --- World Health Organization African Region, 2008 [16]
6
II.2) ETIOLOGIES AND RISK FACTORS OF BACTERIAL
MENINGITIS IN CHILDREN
In 2000, Hib and S. pneumoniae infections are
accountered for approximately 500,000 deaths in the sub-Saharan region.N.
meningitidis has been responsible for recurring epidemics resulting in
700,000 cases of meningitis [17]
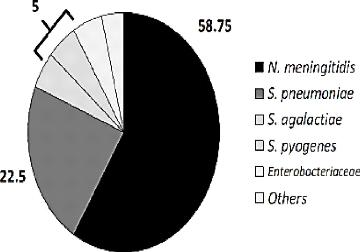
Figure 3: Causes of confirmed bacterial meningitis
from eleven years of active surveillance in a Mexican hospital, 2005 -2016.
[18]
? STREPTOCOCCUS PNEUMONIAE
Streptococcus pneumoniae is one of the main causing
agents responsible for meningitis in newborns, in young children and teenagers
with higher rates of lethality and morbidity [19] [20].
Streptococcus pneumoniae is a Gram - positive, encapsulated bacterium
often found as a normal commensal in the nasopharynx of healthy children
[20].Streptococcus pneumoniae was the commonest cause of
bacterial meningitis in US and Europe, and tends to occur mostly among children
older than 5 years of age [10]. However, the highest risk of
bacterial meningitis caused by Streptococcus pneumoniae
7
is in children greater than 2 years [21]. The
bacterium can become pathogenic, with invasive disease, greatest in patients
who develop meningitis.
? VIRULENCE FACTOR AND PATHOGENESIS
The bacterium is spread by the respiratory fluids from the
infected person when they cough or sneeze, the bacterium then finds its way in
the system where it escapes to the local host defenses and phagocytic
mechanisms, then penetrates the CSF either through choroid plexus /
subarachnoid space originating from bacteremia or via direct extension from
local respiratory system infections [20]. It is able to escape
into the central nervous system easily with the aid of pneumococcal proteins
which include [22]:
> Pneumococcal surface protein A (PspA)
:It is located in the cell wall of the bacterium and acts as a
protective antigen against the host complement system.[22]
> Hyaluronate lyase (Hyl) :This enzyme
mediates facilitation of tissue invasion by breaking down the extracellular
matrix component of the host cell ,thereby increasing tissue permeability.This
factor aids in the pathogenesis of wound infection, meningitis and even
pneumonia[22].
> Autolysin (LytA) :These enzymes are
located in the cell envelope and has a very important role in cell wall
degradation which leads to cell death .They degrade the peptidoglycan backbone
of bacterial organisms , which leads to cell lysis[22].
> Pneumococcal surface antigen A (PsaA) :
This protein is thought to have protective properties and is anchored
to S.pneumoniae through bacterial cell membrane[22].
> Choline binding protein A (CbpA) : It
serves as an anchoring device to pneumococci lipoteichoic C acid structures
present on the surface of the bacterium.Thus aids in the adherence and host
tissue colonization[22].
> Neuraminidase: They enhance colonization
due to their action on gylcans where , they cleave terminal sialic acid from
cell surface gylcans such as mucin, glycolipids and glycoproteins which is
probably responsible for damage to host cell gylcan[22].
Children with basilar skull fractures with CSF leak, asplenism
and HIV infection are at particular risk of developing pneumococcal meningitis
[23], pneumococcal conjugate vaccines have been implemented in
many countries, and immunization with the heptavalent pneumococcal vaccins PCV7
has decreased incidence by incriminating
8
pathogen by greater than 90% [24]. Meanwhile
pneumococcal population undergoes temporal changes in clonal distributions in
the absence of pressure from a vaccine [24].
? HAEMOPHILUS INFLUENZAE
Haemophilus influenzae is Gram-negative coccobacilli
capable of causing serious invasive disease in the child of less than 5 years
of age (Figure 3). Haemophilus influenzae encapsulated serotypes are:
a, b, c, d, e, and f which facilitates its penetration in the blood with the
serotype b being the most virulent of all. The pathogen does not stay alive for
a long time in the environment, it thus has a 12 hours' survival on plastic
objects [25].Haemophilus influenzae type b was the
most common cause of life threatening infection in children in industrialized
countries until universal immunization, where children of less than 5 year of
age, were the primary host with 39% of nasopharynx colonization, but nowadays,
it is instead older children and adults that are considered to be more
susceptible carriers shifting them to primary host[26][25].
? VIRULENCE FACTOR AND PATHOGENESIS
The transmission of the pathogen is done through droplets from
the respiratory airways, through cough, sneezing, speaking from colonized
person, through saliva, and contaminated objects from respiratory secretions.
Sodium hypochlorite at 1%, ethanol at 70%, formaldehyde, glutaraldehyde has
good efficacy against Haemophilus influenzae
[25].However Hib , though not known to produce toxins, it
has the capacity to invade the host system using the following defense
methods[27] :
? Polysaccharide capsule : It is a very
important virulence factor of encapsulated strains of Haemophilus influenza
strains and it protects the bacterium from host immune functions[27].
? Lipooligosaccaharide (LOS) : A major
component of the outer leaflet of the Gram -negative bacteria outer membrane ,
which mediates interactions between bacteria and the host immune system[27].
Hib apart from using the above defense mechanism also uses ,
particular processes to escape from complement systems such as;
- Phase variation which is a immune evasion strategy during
infection where the outer surface of the bacterium is modified to adapt to
changes in the host environment[27].
9
- Binding of host complement regulatory factors which is
important during colonization and infaction, where these factors block activity
at various step of the complement pathway[27].
Globally, Hib accounts for approxi mately 8-13 million serious
illness annually, including 173.000 cases of meningitis causing 78.000 deaths
[28]. The incidence of bacterial meningitis due to the
pathogen has been experiencing a drop in its incidence in developed
countries[8], in Belgium it was at 0,04/100,000 inhabitants in
2012 and even in developing countries where there is implementation of the
vaccin against the Haemophilus influenza type b, less prevalence was
noticed compare to previous years[25]. Despite its reduction
in the cause of meningitis, its identification and prompt treatment are
essential because of the short incubation period which is 2 - 4 days
[25].World Health Organization recommended the addition of Hib
vaccine to immunization programs , according to national capacities and
priorities, however, uptake in developing countries has remained
slow[26].This is partly due to the uncertainty about the true
disease burden[26].
? NEISSERIA MENINGITIDIS
Meningococcal infections occur worldwide as endemic
disease(Figure 3)[29], and it appears that the occurrence of
invasive meningococcal disease is not solely determined by the introduction of
a new virulent bacterial strain but also by other risk factors determining the
transmission of the pathogens [29][19]. Meningococcal
meningitis occurs when Neisseria meningitidis multiplies on the
meninges and in the CSF [30]. Early recognition of this type
of meningitis is important than in any of the acute infectious diseases
[31].
Neisseria meningitidis is a Gram -negative diplococci
which has 13 serogroups defined by specific polysaccharide designated A, B,C,H,
I, K, L, M,X,Y,Z, 29E, and W135(serogroup D is no longer recognized),but is A,
B,C, W135, X, and Y account for most disease where group A is mostly found in
Sub-Saharan Africa, group B found in the temperate climates and group C occurs
mostly as outbreaks [29][32].
Neisseria meningitidis is found in the oropharynx of
10 % of the population with an annual number of invasive disease cases
worldwide estimated to be atleast 1,2 million with 135,000 deaths related to
invasive meningococcal disease and WHO categorizes countries by risk of
meningococcal disease as follows [32];
10
? High risk: countries with greater than 10 cases /100,000 and
/or =1 epidemic Over last 20 years
? Moderate risk: Countries with 2-10 cases /100,000 population
per year
? Low risk: Countries with less than 2 cases /100.000 populations
per year.
The proportion of cases caused by each serogroup varies by age
group also geographic distribution and epidemic potential differ according to
serogroup.Neisseria meningitidis ends to be present particularly in
children less than 5 years old with estimated 500,000 cases and 50,000 deaths
globally each year [29].The largest burden of meningococcal
disease occurs in the sub-Saharan Africa during dry season with the presence of
dust , winds , cold nights with the upper respiratory tract infections combine
to damage the nasopharyngeal mucosa increasing the risk of the disease which is
transmitted through droplets of respiratory secretions while Invasive disease
developing in a small percentage of carriers is regarded as
emergency[32][33].
? VIRULENCE FACTOR AND PATHOGENESIS
Neisseria meningitidis is a fastidious, encapsulated
aerobic bacterium that colonises host mucosal surface using multiple factors
such as[34] :
> Capsule : It is present in strains that
cause invasive disease ,since it provides resistance to antibody and complement
-mediated killing and inhibits phagocytosis[34].
> Lipolysaccharide (LPS) : Induces the
release of chemokines , reactive oxygen speies and nitric oxide and has a role
in resistnce to other host defense[34].
> Adhesins pili : Initiate binding to
epithelial cells,and facilitate passage through the epithelial mucus layer and
movement over the epithelial surfaces .They also facilitates the uptake of DNA
by meningococci and enable adherence to endothelial cells and
erythrocytes[34].
> Opacity proteins : Opa and Opc (only
expressed in Neisseria meningitidis) while Opa is expressed by both
meningococci and gonococci.They have potential roles in pathogenesis that is
not well understood[34].
> Porins : Por A and Por B are porins
through which small nutrients diffuse to the bacterium and they are also
involved in host cell interactions and they are targets for bactericidal
antibodies.Por A is the main component of vesicle based vaccines and a target
for bactericidal antibodies while Por B insert in membranes and induce Ca 2+
influx and activates TLR2 causing cell death[34].
11
? Iron binding proteins: They enable the
meningococci to acquire iron which is an important growth factor during
colonization and disease[34].
World's Health Organization policy's of epidemic containments
prevents at best 50% of cases, therefore for an effective prevention of
meningococcal meningitis in sub Saharan Africa, there should be a strict and
effective follow up of universal vaccination recommendation, but still more
than half of cases among infants less than 1 year are caused by serogroup B
meningococci for which no vaccins is available. Also serogroup X, previously a
rare cause of sporadic meningitis, has been responsible for outbreaks between
2006 and 2010 in Kenya, Niger, Togo, Uganda, and Burkina Faso, the latter with
1,300 cases among the 6, 732 reported annual cases [9][
32].
II.3) RISK FACTORS ASSOCIATED WITH THE OCCURENCE OF
BACTERIAL MENINGITIS IN CHILDREN
The human infection with meningitis has seasonal variation and
this differs from one country to another [33]. Worldwide
meningitis was estimated to cause 1.73.000 deaths in 2002, most children from
the developing countries [35]. Bacterial meningitis as any
other disease has factors that may be associated to its development, and they
can be preventable or not as follows;
? AGE: The first age group (less than 1year)
occupies the highest number of incidence of the disease which tends to be
higher in developing countries than developed countries. The cause might be due
to the immaturity of immune system, lack in the pre-exposure of the body to the
most incriminated organisms which enhances the memory of the immune system to
fight against the invaders[33].
? GEOGRAPHIC ZONE AND CLIMATE: Bacterial
meningitis is endemic in the sub-Saharan region of Africa, especially in those
countries that are included in the «Meningitis Belt» which is made up
of 26 countries from the Senegal to the West to Ethiopia to the East.
Meningitis in tropical areas occurs in dry season and decrease in periods of
rains, while in temperate regions, the epidemics usually occur during winter
and spring seasons [13][ 33].
? SEX: The male sex has been observed in
various studies to be a risk factor for bacterial meningitis. It is not yet
well understood why males will be more susceptible to getting the disease than
female sex [33].
12
? LOW SOCIOECONOMIC STATE AND CROWDING LIVING
CONDITIONS: These are factors that are mostly seen in developing
countries.Crowdness encourage development of meningitis since most of the
detected pathogens are air transmissible [33][35].
? PASSIVE SMOKING: Children exposed to
smoking are found to get meningitis because, passive smokers tends to harbor a
greater number of bacteria in their throat and nasal passage. Also smoking
plays an important role in diminishing the capacity of epithelial cells
covering the respiratory tract for prevention of acquiring infection in
addition to the prevalence of healthy carrier of pathogens [33][
35].
? RECENT UPPER RESPIRATORY TRACT INFECTION:
This can easily be explained by the route of entrance of the
microorganisms to the brain and those important routes of infection are: Otitis
media, mastoiditis, sinusitis and pneumoniae [33].
? HISTORY OF HEAD INJURY AND BRAIN SURGERY:
It is considered an important risk for development of bacterial
meningitis, because of the proximity of the injury with the central nervous
system [33].
? MALNUTRITION: Malnutrition is a complex
disease that if not well controlled affects every system of the body including
the hematopoietic system, and most of the time complicates with anemia. Anemic
patients are highly susceptible to serious infections such as bacterial
meningitis and can be caused by different etiologies [35]. OTHER
FACTORS ASSOCIATED WITH THE DEVELOPMENT OF BACTERIAL MENINGITIS:
? Bottle feeding[33]
? Compromised immune system[33]
? Splenectomy[33]
? Sickle cell disease[33]
? Inherited family tendency for
meningitis[33]
II.4) PATHOPHYSIOLOGY OF BACTERIAL MENINGITIS
There are conditions required to cause invasive diseases such as :
II.4.1) BACTERIAL INVASION
Bacteria reach the central nervous system either by
hematogenous spread or by contiguity like in the case of neonates and children
where pathogens are acquired from
13
non-sterile maternal genital secretions and from organisms
that colonize the upper respiratory tract respectively[29]
Successful colonization of the nasopharyngeal mucosa depends
on the ability of bacteria to evade host defenses including secretory Ig A and
ciliary clearance mechanisms, and to adhere to mucosal epithelium[29].
Microbial virulence factors include the Ig A protease secreted by Neisseria
meningitidis, Streptococcus pneumoniae and Haemophilus influenzae that
cleave Ig A to an active form. Notably meningococus depends on the binding of
fimbriae on the bacterial cell surface to adhere on epithelial cells, and
non-encapsulated strains of meningococci adhere better than capsulated strains.
As the mucosa has been breached and the intravascular space has been entered,
the pathogen must survive in the circulation in order to penetrate the blood
brain barrier[36]. The principal host defense mechanism is complement although
neutrophil and antibodies are also important(Figure 4). The meningeal pathogens
are all capsulated and this virulence factor of theirs enables them to evade
phagocytosis and bactericidal activity of the complement system. In
Streptococcus pneumoniae infection, the alternative complement pathway
is activated by pneumococcal capsular polysaccharides, where there is direct
cleavage of the C3 which generates C3b which opsonizes the organism, enhancing
phagocytic clearance from the circulation[37]. The C3b then binds to Factor B
on the pneumococcal capsular surface offering resistance to opsonisation.
Therefore, it is understandable why individuals with impaired complement
systems are at high risk of getting all the manifestations of invasive
pneumococcal disease.
Neisseria meningitidis, has its capsular sialic acids
which facilitates binding to the C3b to the complement regulatory protein
Factor H, thus blocking activation of the alternative pathway by presenting the
binding of C3b to factor B[36].
In order to cross the blood brain barrier and to overcome
structures such as tight junctions, meningeal pathogens carry effective
molecular tools. They cross the blood brain barrier to enter the subarachnoid
space and are aided with the presence of specific surface bacterial proteins
like E. coli proteins IbeA,IbeB and ompA, Streptococcal proteins such
as CbpA which interacts with glycoconjugate receptor of phosphorlcholine with
platelet activating factor (PAF) on the eukaryotic cells and promotes
endocytosis and crossing the blood brain barrier.N. meningitidis
proteins Opc,
14
Opa, PilC, and a Pili protein[36]. Bacteria causing meningitis
in newborns, most importantly group B streptococcal and Escherichia coli are
also well equipped with adhesive proteins allowing them to invade the central
nervous system[37].
| 


